2. Definition of pKa in aqueous media
2.1 pKa and Brønsted-Lowry acids and bases
Let's recall very briefly that, according to Brønsted-Lowry theory, an acid is a substance capable of yielding a proton to another substance which, possessing the property of capturing a proton, is by definition a base. Let HA be the acid studied, dissolved in water. The following equilibrium is established:
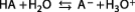

Exclusive to subscribers. 97% yet to be discovered!
Already subscribed? Log in!
Definition of pKa in aqueous media
Article included in this offer
"Characterization and properties of matter"
(
101 articles
)
Updated and enriched with articles validated by our scientific committees
A set of exclusive tools to complement the resources
Bibliography

Exclusive to subscribers. 97% yet to be discovered!
Already subscribed? Log in!

