Overview
ABSTRACT
The direct current electrolysis of aqueous sodium chloride solutions allows for obtaining chlorine, soda and hydrogen. The basic principle of electrolysis cells is to maintain the two gases - chlorine and hydrogen- separated for safety reasons as well as the two products - chorine and soda - which may recombine. Three electrolysis processes are currently used: the "mercury" process, the "diaphragm" process and the "membrane" process.
Read this article from a comprehensive knowledge base, updated and supplemented with articles reviewed by scientific committees.
Read the articleAUTHOR
-
Jean-Christophe MILLET: Process Manager, Chlorochemicals Division - Arkema
INTRODUCTION
The direct current electrolysis of aqueous sodium chloride solutions produces chlorine, soda, and hydrogen. The overall chemical reaction is as follows:
involving two electrons.
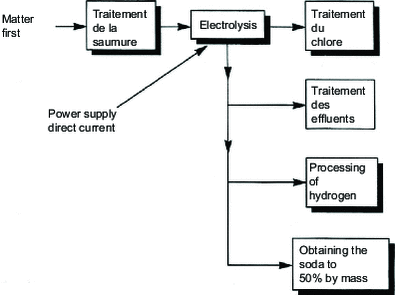
The general principle of the process requires five main sections:
brine purification treatment;
the electrolysis room;
chlorine treatment;
obtaining soda ash at 50% by mass;
hydrogen processing;
and can be represented by the schematic diagram in Figure 1 .
The basic principle of electrolysis cells is to keep the two gases (chlorine and hydrogen) separate for safety reasons (violent reaction between the two gases) and the two products (chlorine and soda) that are likely to recombine.
There are three different electrolysis processes currently in use:
the first with a mercury cathode, known as the "mercury" process;
the second with a percolating diaphragm separating the anode and cathode chambers, known as the "diaphragm" process;
the third with an ion exchange membrane as a separator between the anode and cathode chambers, known as the "membrane" process.

Exclusive to subscribers. 97% yet to be discovered!
Already subscribed? Log in!
EDITIONS
Other editions of this article are available:
Chlorine (Cl2)
Article included in this offer
"Unit operations. Chemical reaction engineering"
(
343 articles
)
Updated and enriched with articles validated by our scientific committees
A set of exclusive tools to complement the resources
Bibliographic references

Exclusive to subscribers. 97% yet to be discovered!
Already subscribed? Log in!

